Recommendations for Fully Vaccinated People
COVID-19 Homepage
COVIDView Summary ending on April 25, 2020
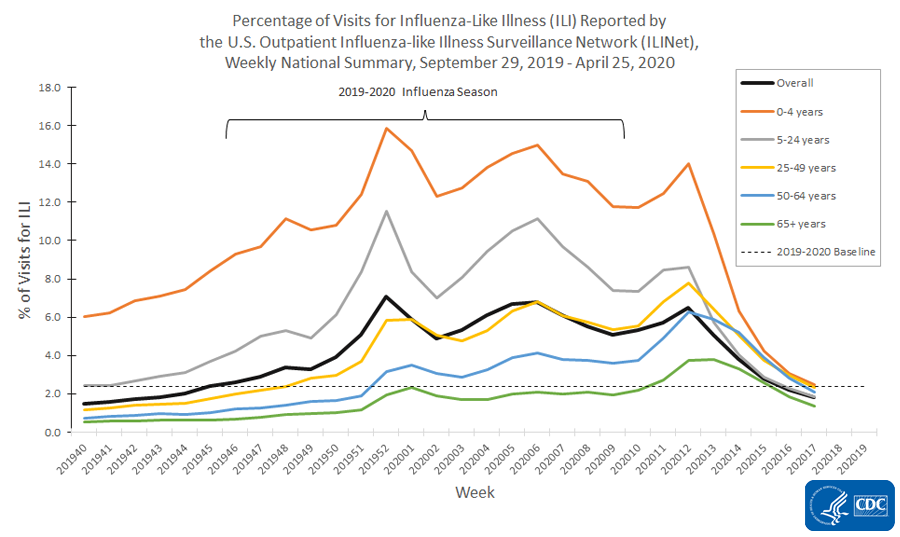
Nationally, levels of influenza-like illness (ILI) declined again this week. They have been below the national baseline for two weeks but remain elevated in the northeastern and northwestern part of the country. Levels of laboratory confirmed SARS-CoV-2 activity remained similar or decreased compared to last week. Mortality attributed to COVID-19 decreased compared to last week but remains significantly elevated and may increase as additional death certificates are counted.
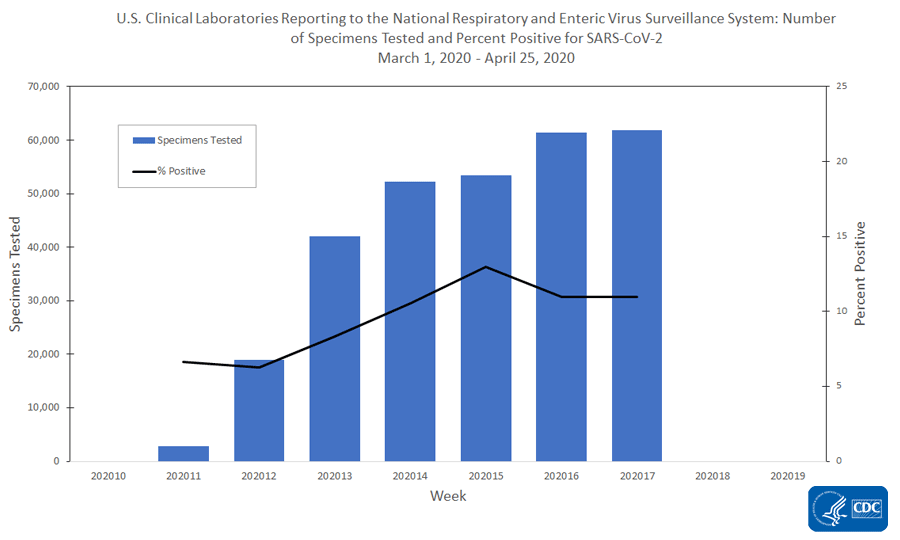
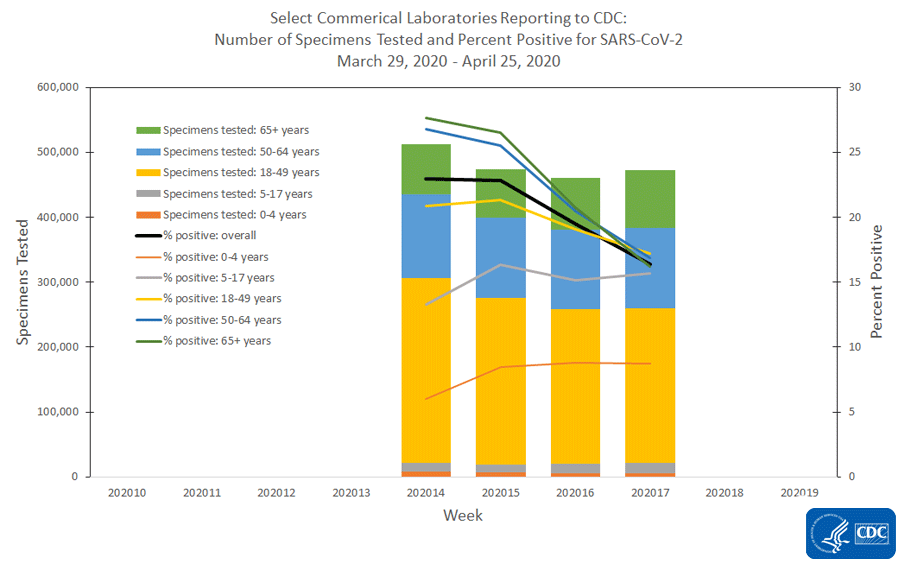
The national percentage of respiratory specimens testing positive for SARS-CoV-2 at public health, clinical and commercial laboratories remained similar or decreased from week 16 to week 17. Percentages by type of laboratory:
- Public health laboratories – decreased from 19.4% during week 16 to 17.1% during week 17;
- Clinical laboratories – remained similar with 10.9% during week 16 and 11.0% during week 17;
- Commercial laboratories – decreased from 19.5% during week 16 to 16.4% during week 17.
Two indicators from existing surveillance systems are being used to track outpatient or emergency department (ED) visits for illness with symptoms compatible with COVID-19.
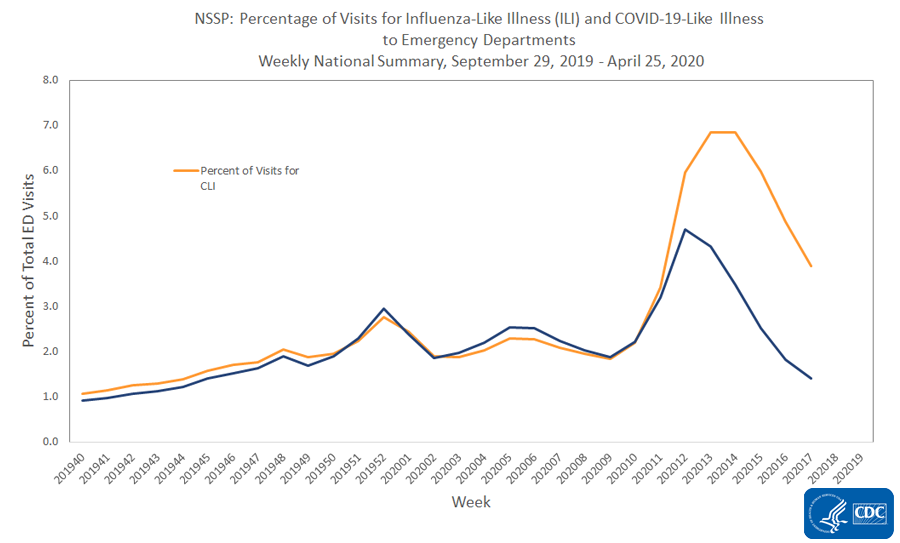
- Nationally, the percentages of visits for ILI and COVID-19-like illness (CLI) decreased compared to last week. Levels of ILI are now below baseline for the second week.
Recent changes in health care seeking behavior are likely affecting data reported from both networks, making it difficult to draw further conclusions at this time. Tracking these systems moving forward will give additional insight into illness related to COVID-19.
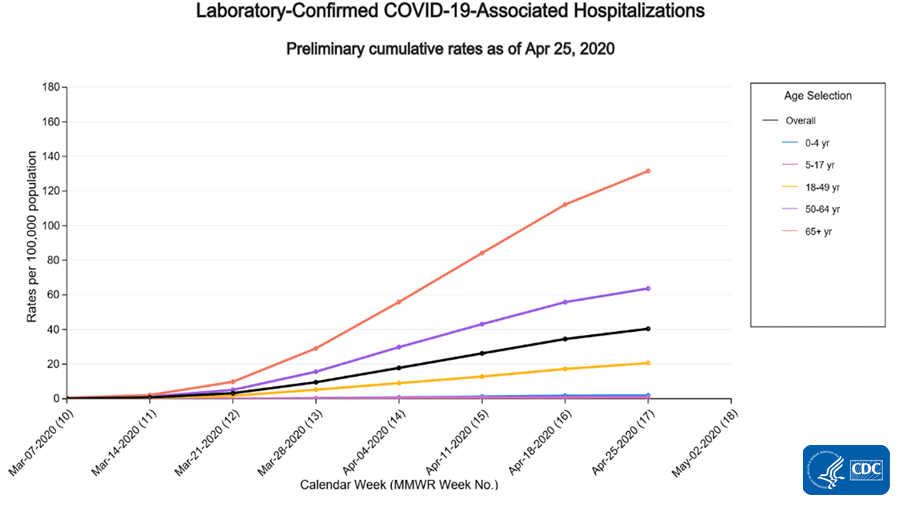
Cumulative COVID-19-associated hospitalization rates since March 1, 2020, are updated weekly. The overall cumulative hospitalization rate is 40.4 per 100,000, with the highest rates in people 65 years and older (131.6 per 100,000) and 50-64 years (63.7 per 100,000).
Based on death certificate data, the percentage of deaths attributed to pneumonia, influenza or COVID-19 (PIC) decreased from 23.6% during week 16 to 14.6% during week 17 but remained significantly above baseline. This is the second week of declines in this indicator, but this percentage may change as death certificates representing recent deaths are processed.
All data are preliminary and may change as more reports are received.
A description of the surveillance systems summarized in COVIDView, including methodology and detailed descriptions of each data component, is available on the surveillance methods page.
- Nationally, the percentage of laboratory specimens testing positive for SARS-CoV-2 remained similar, or decreased, compared to last week.
- Nationally, visits to outpatient providers and emergency departments (EDs) for illnesses with symptoms consistent with COVID-19 continued to decline and are below baseline nationally and in many regions of the country. They remain elevated in the northeast and northwest.
- The decrease in the percentage of people presenting for care with ILI and CLI may be due to a decline in COVID-19 illness. Reported levels of activity may be decreasing because of widespread adoption of social distancing efforts and changes in healthcare seeking behavior.
- Little influenza virus activity has been reported in recent weeks.
- The overall cumulative COVID-19 associated hospitalization rate is 40.4 per 100,000, with the highest rates in people 65 years and older (131.6 per 100,000) and 50-64 years (63.7 per 100,000).
- Hospitalization rates for COVID-19 in adults (18-64 years) are higher than hospitalization rates for influenza at comparable time points* during the past 5 influenza seasons.
- For people 65 years and older, current COVID-19 hospitalization rates are similar to those observed during comparable time points* during recent high severity influenza seasons.
- For children (0-17 years), COVID-19 hospitalization rates are much lower than influenza hospitalization rates during recent influenza seasons.
- Based on death certificate data, the percentage of deaths attributed to pneumonia, influenza or COVID-19 (PIC) decreased from 23.6% during week 16 to 14.6% during week 17 which is still significantly above baseline. This is the second week of decline in this indicator, but the percentage remains high compared with any influenza season. The percentage may change as additional death certificates for deaths during recent weeks are processed.
- Declines in some key indicators used to track COVID-19 from one week to the next could change as additional data are received but also may be a result of widespread social distancing measures.
*Number of weeks since 10% of specimens tested positive for SARS-CoV-2 and influenza, respectively.
The number of specimens tested for SARS-CoV-2 and reported to CDC by public health laboratories and a subset of clinical and commercial laboratories in the United States are summarized below. At this point in the outbreak, all laboratories are performing primary diagnostic functions; therefore, the percentage of specimens testing positive across laboratory types can be used to monitor trends in COVID-19 activity. As the outbreak progresses, it is possible that different types of laboratories will take on different roles, and the data interpretation may need to change. The lower percentage of specimens testing positive in clinical laboratories compared to public health and commercial laboratories may be influenced by COVID-19 activity in areas with reporting laboratories and by larger proportions of specimens obtained from children tested in these laboratories.
| Week 17 (April 19 – April 25, 2020) |
Cumulative since March 1, 2020 | |
|---|---|---|
| No. of specimens tested | 701,913 | 3,906,678 |
| Public Health Laboratories | 110,603 | 523,380 |
| Clinical Laboratories | 61,818 | 292,756 |
| Commercial Laboratories | 529,492 | 3,090,542 |
| No. of positive specimens (%) | 112,298 (16.0%) | 702,814 (18.0%) |
| Public Health Laboratories | 18,891 (17.1%) | 85,524 (16.3%) |
| Clinical Laboratories | 6,784 (11.0%) | 30,816 (10.5%) |
| Commercial Laboratories | 86,623 (16.4%) | 586,474 (19.0%) |
* Commercial and clinical laboratory data represents select laboratories and does not capture all tests performed in the United States.
Public Health Laboratories
Clinical Laboratories
Commercial Laboratories
* Commercial laboratories began testing for SARS-CoV-2 in early March, but the number and geographic distribution of reporting commercial laboratories became stable enough to calculate a weekly percentage of specimens testing positive as of March 29, 2020.
Additional virologic surveillance information: Surveillance Methods
Two syndromic surveillance systems are being used to monitor trends in outpatient and emergency department visits that may be associated with COVID-19. Each system monitors a slightly different syndrome, and together these systems provide a more comprehensive picture of mild to moderate COVID-19 illness than either would individually. Both systems are currently being affected by recent changes in healthcare seeking behavior, including increasing use of telemedicine, recommendations to limit emergency department (ED) visits to severe illnesses, and increased practice of social distancing. These changes affect the numbers of people and their reasons for seeking care in the outpatient and ED settings.
ILINet
The U.S. Outpatient Influenza-like Illness Surveillance Network (ILINet) provides data on visits for influenza-like illness (ILI) (fever [≥100○F] and cough and/or sore throat) to approximately 2,600 primary care providers, emergency departments and urgent care centers in all 50 states, Puerto Rico, the District of Columbia and the U.S. Virgin Islands. Mild COVID-19 illness presents with symptoms similar to ILI, so ILINet is being used to track trends of mild to moderate COVID-19 illness and allows for comparison with prior influenza seasons.
Nationwide during week 17, 1.8% of patient visits reported through ILINet were due to ILI. This percentage is below the national baseline of 2.4% and represents the fifth week of a decline after three weeks of increase beginning in early March. The percentage of visits for ILI decreased in all age groups. Nationally, laboratory-confirmed influenza activity as reported by clinical laboratories has decreased to levels usually seen in summer months which, along with changes in healthcare seeking behavior and the impact of social distancing, is likely contributing to the decrease in ILI activity.
* Age-group specific percentages should not be compared to the national baseline.
On a regional levelExternal, the percentage of outpatient visits for ILI ranged from 0.9% to 3.7% during week 17. Compared to week 16, the percent of outpatient visits for ILI increased slightly in region 5, but decreased in all other regions, and six regions are below their region-specific baselines.
ILI Activity Levels
Data collected in ILINet are used to produce a measure of ILI activity for all 50 states, Puerto Rico, the District of Columbia and New York City. The mean reported percentage of visits due to ILI for the current week is compared to the mean reported during non-influenza weeks, and the activity levels correspond to the number of standard deviations below, at or above the mean.
The number of jurisdictions at each activity level during week 17 and the change compared to the previous week are summarized in the table below and shown in the following maps. The decreasing percentage of visits for ILI described above are reflected in this week’s ILI activity levels.
| Activity Level | Number of Jurisdictions | |
| Week 17 (Week ending April 25, 2020) |
Compared to Previous Week | |
| Very High | 0 | -1 |
| High | 3 | -4 |
| Moderate | 8 | +6 |
| Low | 2 | -7 |
| Minimal | 40 | +6 |
| Insufficient Data* | 1 | No change |
*Data collected in ILINet may disproportionally represent certain populations within a state, and may not accurately depict the full picture of influenza activity for the whole state. Differences in the data presented here by CDC and independently by some state health departments likely represent differing levels of data completeness with data presented by the state likely being the more complete.
National Syndromic Surveillance Program (NSSP): Emergency Department (ED) Visits
NSSP is a collaboration among CDC, federal partners, local and state health departments and academic and private sector partners to collect, analyze and share electronic patient encounter data received from multiple healthcare settings. To track trends of potential COVID-19 visits, visits for COVID-19-like illness (CLI) (fever and cough or shortness of breath or difficulty breathing or presence of a coronavirus diagnosis code) and ILI to a subset of emergency departments in 47 states are being monitored.
Nationwide during week 17, 3.9% of emergency department visits captured in NSSP were due to CLI and 1.4% were due to ILI. This is the fifth week of decline in percentage of visits for ILI and the fourth week of stable or declining percentage of visits for CLI. All 10 HHS regionsExternal experienced a decline in percentage of visits for ILI and CLI.
Additional information about medically attended outpatient and emergency department visits for ILI and CLI: Surveillance Methods
The COVID-19-Associated Hospitalization Surveillance Network (COVID-NET) conducts population-based surveillance for laboratory-confirmed COVID-19-associated hospitalizations in select counties participating in the Emerging Infections Program (EIP) and states participating in the Influenza Hospitalization Surveillance Project (IHSP). COVID-NET-estimated hospitalization rates are updated weekly.
A total of 13,114 laboratory-confirmed COVID-19-associated hospitalizations were reported by sites between March 1, 2020, and April 25, 2020. The overall cumulative hospitalization rate was 40.4 per 100,000 population. The highest rate of hospitalization is among adults aged ≥ 65 (131.6 per 100,000), followed by adults aged 50-64 years (63.7 per 100,000) and adults aged 18-49 years (20.6 per 100,000).
Among 4,384 cases with information on race/ethnicity, 40.2% were non-Hispanic white, 36.4% were non-Hispanic black, 12.2% were Hispanic, and 11.2% were other race, including unknown race.
| Overall | 0-4 years | 5-17 years | 18-49 years | 50-64 years | 65+ years | |
| N (%) | N (%) | N (%) | N (%) | N (%) | N (%) | |
| Non-Hispanic White | 1,761 (40.2) | 4 (36.4) | 5 (27.8) | 234 (23.3) | 461 (34.0) | 1,057 (53.0) |
| Non-Hispanic Black | 1,595 (36.4) | 2 (18.2) | 7 (38.9) | 386 (38.4) | 546 (40.3) | 654 (32.8) |
| Hispanic | 536 (12.2) | 3 (27.3) | 5 (27.8) | 253 (25.2) | 177 (13.1) | 98 (4.9) |
| Other | 491 (11.2) | 2 (18.2) | 1 (5.6) | 132 (13.1) | 170 (12.6) | 186 (9.3) |
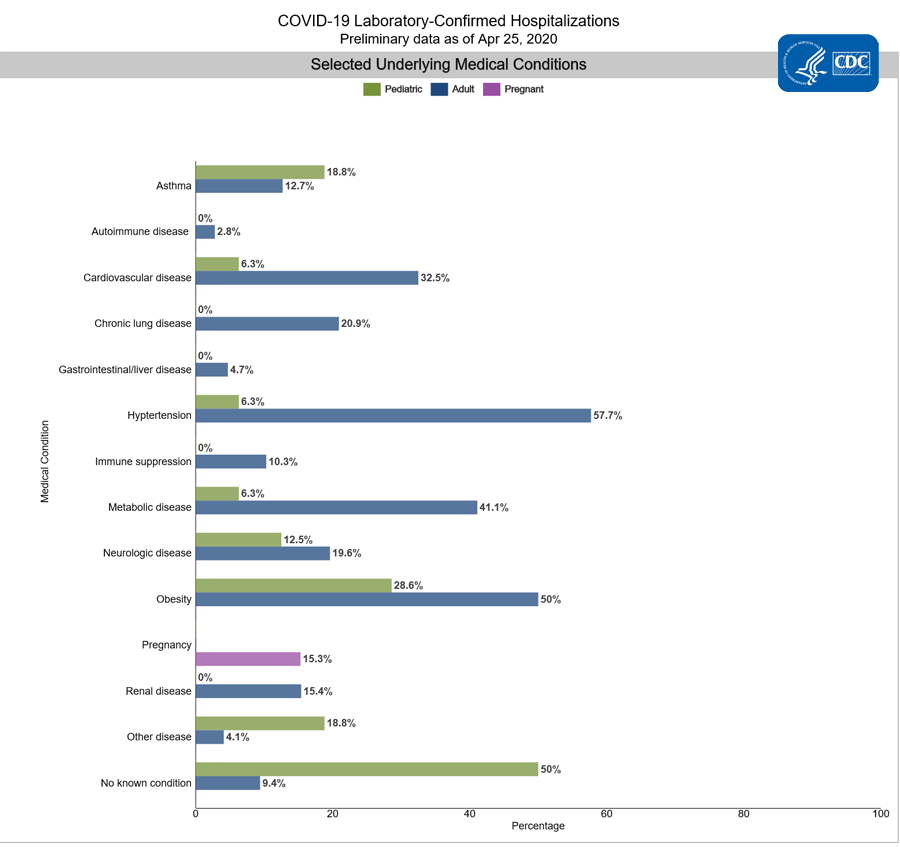
Among 2,028 hospitalized adults with information on underlying medical conditions, 90.6% had at least one reported underlying medical condition, the most commonly reported were hypertension, obesity, chronic metabolic disease, and cardiovascular disease.
Additional hospitalization surveillance information: Surveillance Methods | Additional rate data | Additional demographic and clinical data
The National Center for Health Statistics (NCHS) collects death certificate data from vital statistics offices for all deaths occurring in the United States. Based on death certificate data available on April 30, 2020, 14.6% of all deaths occurring during the week ending April 25, 2020 (week 17) were due to pneumonia, influenza or COVID-19 (PIC). This is the second week of decline; however, the percentage remains significantly above the epidemic threshold of 6.8% for week 17. Data for week 17 are incomplete, and the PIC percentage may increase as more death certificates representing deaths during week 17 are filed.
*Data during recent weeks are incomplete because of the lag in time between when the death occurred and when the death certificate is completed, submitted to NCHS and processed for reporting purposes.
Additional NCHS mortality surveillance information: Surveillance Methods | Provisional Death Counts for COVID-19